Compound: Bacteriostatic Water
CAS Number: 7732-18-5
Molecular Formula: H2O + 0.9% Benzyl Alcohol
Purity: N/A
Third-Party Testing: Janoshik Analytical (independent lab, Prague)
COA Verification: janoshik.com/verify
Batch: MRL-BAC-0326
Source: Canadian manufactured
Shipping: Same-day from Vancouver, BC
Bacteriostatic water Canada: pharmaceutical-grade sterile water containing 0.9% benzyl alcohol as a bacteriostatic preservative, designed for research peptide reconstitution. Available from Maple Research Labs in 30ml vials for laboratory use.
Product Specifications
- Volume: 30ml per vial
- Preservative: 0.9% Benzyl Alcohol (USP grade)
- Base: Sterile Water for Injection (USP grade)
- Sterility: Sterile-filtered, 0.22 micron
- Container: Multi-use glass vial with rubber stopper
- pH: 5.0-7.0
Bacteriostatic Water Canada Overview
Bacteriostatic water (BAC water) is sterile water that has been preserved with 0.9% benzyl alcohol, a bacteriostatic agent that inhibits microbial growth and allows the solution to be used over multiple withdrawals from the same vial. This bacteriostatic water Canada product is manufactured to pharmaceutical-grade standards for use in research peptide reconstitution protocols.
Unlike sterile water for injection (SWFI), which must be used immediately after opening and discarded after a single withdrawal, bacteriostatic water can be stored and reused for up to 28 days after initial penetration of the vial stopper. This multi-use capability makes it the standard reconstitution vehicle for lyophilized research peptides that will be used across multiple experimental sessions.
Benzyl Alcohol Preservation Mechanism
The 0.9% benzyl alcohol concentration acts as a bacteriostatic (growth-inhibiting) rather than bactericidal (growth-killing) preservative. Benzyl alcohol disrupts microbial membrane integrity and interferes with microbial metabolic processes at concentrations sufficient to prevent colony growth but below cytotoxic thresholds for mammalian cells in most research applications.
The 0.9% concentration represents the standard pharmaceutical formulation established in the United States Pharmacopeia (USP). This concentration has been validated to maintain sterility over the 28-day multi-use period when proper aseptic withdrawal technique is followed in laboratory settings.
Research Peptide Reconstitution
Lyophilized research peptides require reconstitution in an appropriate aqueous vehicle before use in experimental protocols. Bacteriostatic water is the preferred reconstitution vehicle for peptides that will be stored and used over multiple days or weeks. The benzyl alcohol preservative prevents bacterial contamination that could degrade the peptide or introduce confounding variables in research assays.
For peptide reconstitution, the bacteriostatic water should be added slowly to the lyophilized peptide vial, directed against the glass wall rather than directly onto the peptide cake. Gentle swirling rather than vortexing is recommended to avoid peptide denaturation from shear forces. Once reconstituted, peptide solutions should be stored at 2-8C and used within the timeframe appropriate for each specific peptide.
Some research protocols require preservative-free sterile water rather than bacteriostatic water. Researchers should verify which reconstitution vehicle is appropriate for their specific assay system, as benzyl alcohol may interfere with certain cell culture or bioassay protocols.
Bacteriostatic Water Canada Quality Standards
Every batch of bacteriostatic water Canada from Maple Research Labs meets pharmaceutical-grade standards for sterility and preservative concentration. Each production lot undergoes sterility testing, endotoxin screening, and benzyl alcohol concentration verification to confirm the 0.9% USP specification. Our quality assurance ensures consistent sterile water quality for reliable peptide reconstitution across all shipments.
Storage and Handling
Store unopened vials at room temperature (15-30C), protected from light. After first use, store at 2-8C. Discard 28 days after initial stopper penetration. Use aseptic technique for all withdrawals.
Shipping
Ships same-day from our Canadian facility. Packaged to prevent breakage during transit.
For research and laboratory use only. Not for injection or human administration.
Related Research Articles
Learn more about research supplies and peptide handling:
- Shipping Research Peptides Within Canada: What to Expect
- Research Peptide Supply Chain: From Synthesis to Laboratory
- Certificates of Analysis (COA) Documentation
- Browse All Research Products
Frequently Asked Questions
What is bacteriostatic water?
Bacteriostatic water is sterile water containing 0.9% benzyl alcohol as a preservative. The benzyl alcohol inhibits microbial growth, allowing the water to be used for multiple reconstitutions from the same vial.
How long does bacteriostatic water last after opening?
Bacteriostatic water maintains preservative efficacy for up to 28 days after first puncture when stored at 2-8C. After 28 days, the benzyl alcohol concentration may be insufficient to prevent microbial contamination.
What is bacteriostatic water used for in research?
It is the standard reconstitution solvent for lyophilized research peptides. The preservative allows multi-access use, unlike sterile water which should be used within 24 hours.
 Maple
Maple 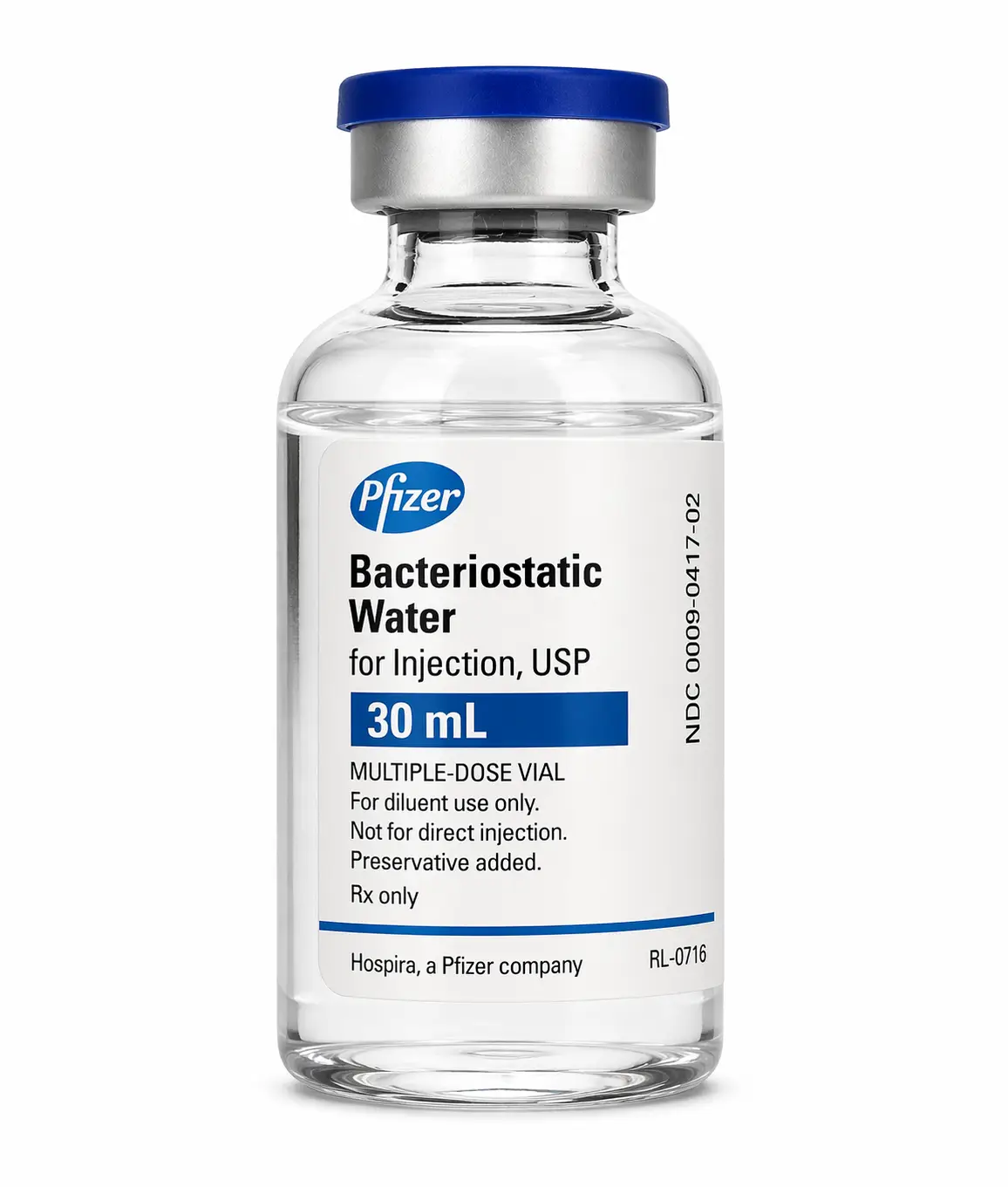




Reviews
There are no reviews yet.